 Notably, the observed efficacy of both vaccines one year after vaccination supports the implementation of an early life SARS-CoV-2 vaccine. Overall, the Protein+3 M-052-SE vaccine provided superior protection to the mRNA-LNP vaccine, emphasizing opportunities for optimization of current vaccine platforms. Neutralizing and binding antibody responses to the B.1.617.2 variant at the day of challenge correlated with lung pathology and reduced virus replication. In contrast, vaccinated rhesus macaques had faster viral clearance with mild to no pneumonia. Seven of eight control rhesus macaques exhibited severe interstitial pneumonia and high virus replication in the upper and lower respiratory tract. At one year, corresponding to human toddler age, we challenged vaccinated rhesus macaques and age-matched non-vaccinated controls intranasally and intratracheally with a high-dose of heterologous SARS-CoV-2 B.1.617.2 (Delta). Here, we demonstrate that broadly neutralizing and spike-binding antibodies against variants of concern (VOC), as well as T cell responses, persisted for 12 months. 
We demonstrated previously that a two-dose regimen of stabilized prefusion Washington SARS-CoV-2 S-2P spike (S) protein encoded by mRNA encapsulated in lipid nanoparticles (mRNA-LNP) or purified S-2P mixed with 3 M-052, a synthetic toll-like receptor (TLR) 7/8 agonist, in a squalene emulsion (Protein+3 M-052-SE) was safe and immunogenic in infant rhesus macaques. Yet, questions regarding the durability of vaccine efficacy, especially against emerging variants, in this age group remain. Food and Drug Administration only gave emergency-use-authorization of the BNT162b2 and the mRNA-1273 SARS-CoV-2 vaccines for infants 6 months and older in June 2022. , Glicoproteínas de Hemaglutininação de Vírus da Influenza Our results support this platform as a step forward in the development of a universal influenza vaccine. These responses were durable, with neutralizing antibodies observed more than 1 year after vaccination. We found that H1ssF elicited cross-reactive neutralizing antibodies against the conserved HA stem of group 1 influenza viruses, despite previous H1 subtype head-specific immunity. The most common symptoms included pain or tenderness at the injection site (n = 10, 19%), headache (n = 10, 19%), and malaise (n = 6, 12%). H1ssF was safe and well tolerated, with mild solicited local and systemic reactogenicity. The primary objective of this trial was to evaluate the safety and tolerability of H1ssF, and the secondary objective was to evaluate antibody responses after vaccination. Thirty-five (74%) 60-µg dose participants received the boost, whereas 11 (23%) boost vaccinations were missed because of public health restrictions in the early stages of the COVID-19 pandemic. Fifty-two healthy adults aged 18 to 70 years old enrolled to receive either 20 µg of H1ssF once (n = 5) or 60 µg of H1ssF twice (n = 47) with a prime-boost interval of 16 weeks. This first-in-human dose-escalation open-label phase 1 clinical trial (NCT03814720) tested an HA stabilized stem ferritin nanoparticle vaccine (H1ssF) based on the H1 HA stem of A/New Caledonia/20/1999. A vaccine without the variable HA head domain has the potential to focus the immune response on the conserved HA stem. Immunodominance of the influenza hemagglutinin (HA) head in currently licensed vaccines impedes induction of cross-reactive neutralizing stem-directed antibodies.  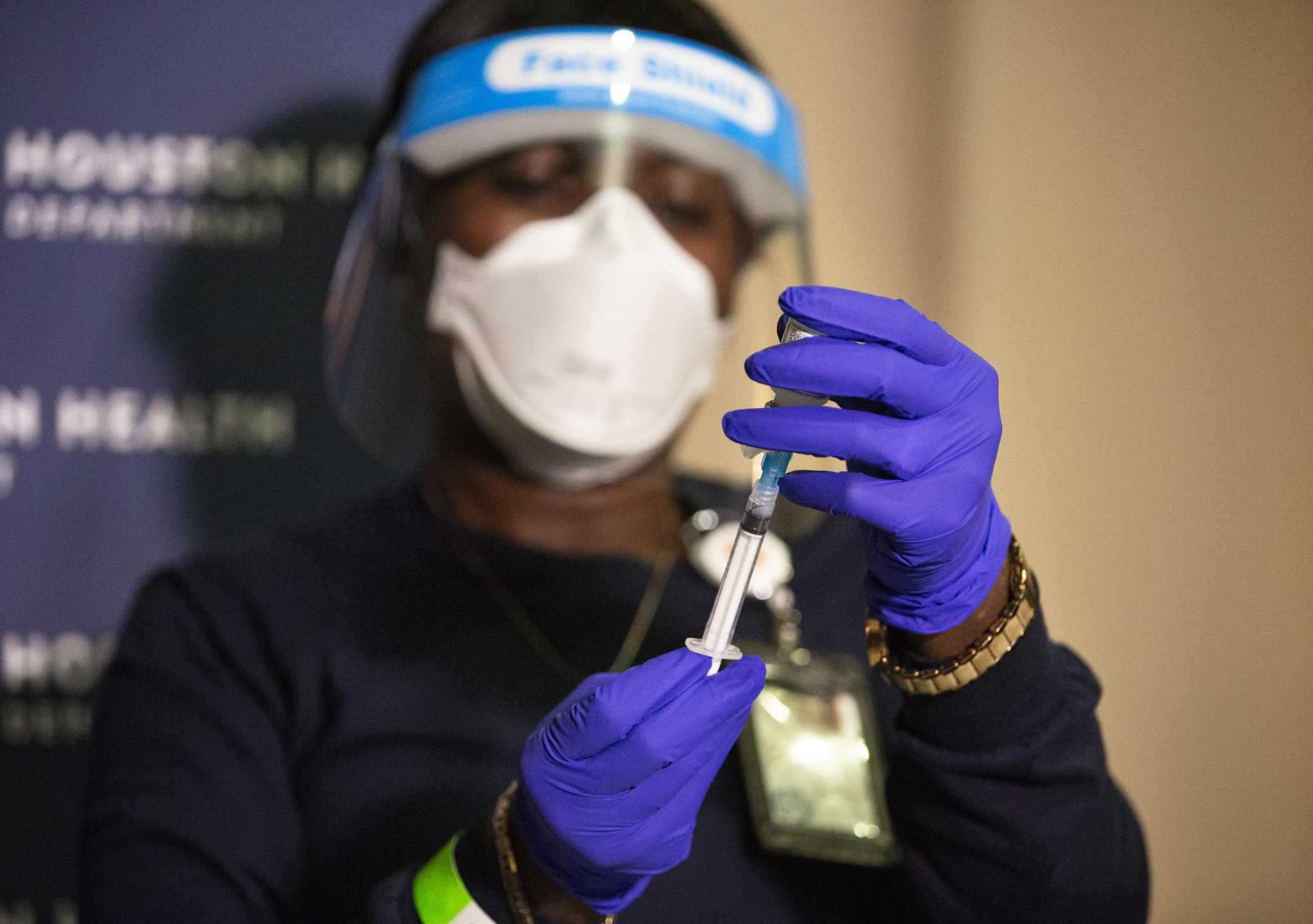 
Influenza vaccines could be improved by platforms inducing cross-reactive immunity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
